Determination of Hydrogen Cyanide in Mainstream Tobacco Smoke
Health Canada
T-107 December 31, 1999
Table of Contents
- Scope of Applications
- Normative references
- Definitions
- Method Summary
- Apparatus and Equipment
- Reagents and Supplies
- Preparation of Glassware
- Preparation of Solutions
- Preparation of Standards
- Sampling
- Tobacco Product Preparation
- Smoking Machine Preparation
- Sample Generation
- Sample Analysis
- Quality Control
- Modifications for Intense Smoking Conditions
- Reference
- Appendix
1 Scope of Applications
- Applicable to the quantitation of the hydrogen cyanide (HCN) content of mainstream tobacco smoke by an automated continuous flow analyzer, as trapped on the glass fibre filter disc (pad) and in the gas phase.
2 Normative References
- American Society for Testing and Materials (ASTM) D 1193-77 - Standard Specifications for Reagent Water, Version 1977.
- Health Canada Test Method T-115 - Determination of Tar, Water, Nicotine and Carbon Monoxide in Mainstream Tobacco Smoke, 1999-12-31.
3 Definitions
- Refer to T-115 for definitions of terms used in this document.
4 Summary of Method
- Five conditioned cigarettes* are smoked per port, on alternate ports of an automated linear constant volume smoking machine, onto a conditioned pad, with a trap containing 0.1N NaOH located directly behind the pad.
*For other tobacco products, select a number such that breakthrough does not occur. - The pad is extracted with 40 mL of 0.1N NaOH on a wrist action shaker for 30 minutes.
- Both the pad extract and impinger trapping solutions are analyzed by an automated continuous flow colorimetric analyzer where each sample undergoes on-line dilution. Hydrogen cyanide in the sample is converted to cyanogen chloride by an aqueous
solution of chloramine-T. The cyanogen chloride then reacts with pyridine to give glutaconic aldehyde, which, upon reaction with a pyrazolone reagent, forms a coloured complex. A single channel monitors the complex, which is quantified
by comparison to an external standard calibration.
Note: The testing and evaluation of certain products against this test method may require the use of materials and or equipment that could potentially be hazardous and this document does not purport to address all the safety aspects associated with its use. Anyone using this test method has the responsibility to consult with the appropriate authorities and to establish health and safety practices in conjunction with any existing applicable regulatory requirements prior to its use.
The analysis should be completed in one day, and the waste potassium cyanide solutions generated must be stored for disposal by registered chemical recycling agencies. All pipetting must be done with mechanical devices.
5 Apparatus and Equipment
- Equipment needed to perform conditioning as specified in T-115.
- Equipment needed to perform marking for butt length as specified in T-115
- Equipment needed to perform smoking of tobacco products as specified in T-115
- Analytical Balance capable of reading to four decimal places.
- 125 mL Polymethylpentene (PMP) Erlenmeyer flasks with screw caps.
- Wrist-action shaker.
- 5 cc Disposable Syringe.
- Syringe Filter - Nalgene SFCA (25 mm) (or equivalent).
- Computer-controlled Continuous Flow AutoAnalyzer (or equivalent) consisting of :
- Technicon IV Autosampler.
- Technicon II Peristaltic Pump.
- HCN Manifold.
- Single Channel Colorimeter equipped with 15 mm flow cell and 540 nm filter.
- 70 mL impingers with coarse frits.
- 50 mL volumetric flasks with ground glass joints.
- 50 mL graduated cylinders.
- Glass filter funnel.
- Magnetic Stirrer and stir bars.
- 1000 μL variable adjusting volume pipettor.
- Sample cups for autoanalyzer.
6 Reagents and Supplies
Note: All reagents shall be, at the least, recognized as analytical reagent grade in quality.
- Potassium cyanide.
- Chloramine T.
- Pyridine.
- Sodium Hydroxide (NaOH).
- 3-methyl-1-phenyl-2-pyrazolin-5-one.
- Bispyrazolone.
- Potassium dihydrogen phosphate.
- Disodium hydrogen phosphate.
- Brij-35 solution (30 %).
- Type I water, as specified in ASTM D1193.
7 Preparation of Glassware
- Glassware should be cleaned and dried in such a manner to ensure that contamination from glassware does not occur.
8 Preparation of Solutions
- Chloramine-T Solution
- Add 2 g of chloramine-T to 500 mL of Type I water. Mix well. Prepare fresh weekly.
- Saturated Pyrazolone Solution
- Stir 5 g of 3-methyl-1-phenyl-2-pyrazolin-5-one with 2 L of water for five hours, using a magnetic stirrer and stir bar.
- Pyridine-Pyrazolone Solution
- Dissolve 0.080 g of bispyrazolone in 80 mL of pyridine in an amber bottle and mix on magnetic stirrer for 30 minutes. After complete solution is obtained, add 400 mL of filtered saturated pyrazolone solution and mix.
- Buffer solution
- Dissolve 13.6 g of potassium dihydrogen phosphate and 0.28 g of disodium hydrogen phosphate in Type I water and dilute to 1 L. Add 0.5 mL of Brij-35 and mix.
- Sodium Hydroxide (0.1N)
- Add 8g of NaOH pellets to 2 L of Type I water. Stir until completely dissolved.
9 Preparation of Standards
- Preparation of Cyanide Standards
- Prepare a primary stock solution equivalent to 500ppm HCN (60.2 mg of KCN made up to 50 mL with 0.1N NaOH).
- Then dilute 0.1, 0.3, 0.5, 0.7, 0.9, 1.2 and 1.5 mL aliquots of this solution each to 50 mL with 0.1N NaOH. These standards are equivalent to 1, 3, 5, 7, 9, 12 and 15ppm (µg/mL) HCN. These standards should be prepared fresh weekly.
10 Sampling
- The sampling of tobacco products for the purpose of testing shall be as specified in T-115.
11 Tobacco Product Preparation
- Product is to be conditioned as specified in T-115.
- Cigarettes, cigarette equivalents, bidis, kreteks and cigars are to be marked for butt length as specified in T-115.
- Cigarettes to be smoked under intense conditions shall be prepared as specified in T-115.
12 Smoking Machine Preparation
- Ambient Conditions
- The ambient conditions for smoking shall be as those specified in T-115
- Machine Conditions
- The machine conditions shall be as those specified in T-115 with the following modifications:
- Load impingers for the vapour phase HCN analysis with 30 mL of 0.1N NaOH. Place the impingers onto the rear section of the smoking machine. Tubing from the top of the impinger (internal stem connection) attaches to the pad holder and tubing from the side of the impinger attaches to the vacuum source of system (syringe).
- The machine conditions shall be as those specified in T-115 with the following modifications:
13 Sample Generation
- Cigarettes shall be smoked and TPM collected as specified in T-115.
14 Sample Analysis
- Extraction of Pads
- Remove the mainstream pad, fold in half and in half again with the "clean" side facing out. Grasp with a pair of clean tweezers, and wipe the holder. Place the pad into a clean labeled 125 mL Erlenmeyer flask.
- Add 40 mL of 0.1N NaOH to each of the Erlenmeyer flasks containing a pad from the analytical run and cap.
- Clamp the flasks onto the armature of a wrist action shaker and agitate for 30 minutes. The pad should be disintegrated at the end of the time period.
- Filter the pad extract directly into appropriately labeled vials or sample cups using a syringe filter attached to a 5 cc disposable syringe.
- Analyse the extract immediately for HCN (within 24 hours).
- Impinger Trapping Solution
- The 0.1N NaOH impinger solution that traps the vapour phase HCN is used to rinse the attached tubes and walls of the impinger. The impinger solution is then poured into autosampler cups and analysed for HCN while the pad extracts are shaking.
- Continuous Flow Analysis
- The Autosampler is operated at a sampling rate of 20 per hour with a 2:1 sample to wash ratio. Sufficient time should be allowed for the system to become stable with the reagents being pumped.
- The samples are only rerun if found to be out of range or if there is a problem with the analysis.
- The samples undergo on-line dilution.
- Sampling cups containing only 0.1N NaOH are placed at regular intervals to allow for baseline correction.
- Calculations
- Construct a calibration curve relating ppm of HCN to peak height with the data obtained from the standards.
- Obtain ppm or mg/mL of HCN for each extract and calculate micrograms (mg) per cigarette of HCN in mainstream smoke for both pad and impinger extracts:
Pad HCN (μg/cigarette) = [amount (μg/mL) × 40 (mL)] / No. of Cigarettes smoked
Impinger HCN (μg/cigarette) = [amount (μg/mL) × 30 (mL)] / No. of Cigarettes smoked
Total HCN (μg/cigarette) = PAD HCN (μg/cigarette) + IMPINGER HCN (μg/cigarette)
15 Quality Control
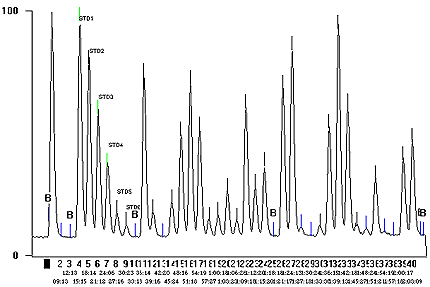
- Typical Chromatogram
- See Appendix 1
- Recoveries and Levels of Contamination
- Each analytical run of test cigarettes should also include:
- A Laboratory Reagent Blank (LRB) to evaluate the extent of any interferences due to glassware, trapping reagents, pads, and analyzer effects.
LRB: Add one conditioned filter pad to a clean 125 mL Erlenmeyer flask, add 40 mL of 0.1N NaOH solution and cap. - A Laboratory Fortified Blank (LFB) to evaluate the extent of potential analyte loss.
LFB: Add one conditioned filter pad to a clean 125 mL Erlenmeyer flask, add 39.6 mL of 0.1N NaOH plus 0.4 mL of the 500 ppm HCN stock solution and cap. - A Laboratory Fortified Matrix (LFM) to evaluate any potential matrix effects.
LFM: After shaking the flasks, prepare a laboratory fortified matrix (LFM) daily using a control brand: LFMA - Dilute 5 mL of a control pad extract to 10 mL with 0.1 N NaOH.
LFMB - Dilute 5 mL of a control pad extract with 0.1 mL of the 500 ppm KCN stock solution and make to 10 mL with 0.1N NaOH. - Check standards run as samples to verify the calculation process and validate the calibration.
- A Laboratory Reagent Blank (LRB) to evaluate the extent of any interferences due to glassware, trapping reagents, pads, and analyzer effects.
- Each analytical run of test cigarettes should also include:
- Method Detection Limit (MDL) & Limit of Quantification (LOQ)
- Method Detection Limit (MDL)
- The method detection limit is determined by analyzing the lowest level standard at least 10 times as an unknown over several days. The MDL is then calculated as three times the standard deviation of these determinations. Typical values are: 0.036 µg/mL, which gives an impinger MDL of 0.22 µg/cigarette and a pad MDL of 0.29 µg/cigarette.
- Limit of Quantification (LOQ)
- The limit of quantification is determined by analyzing the lowest level standard at least 10 times as an unknown over several days. The LOQ is then calculated as 10 times the standard deviation of these determinations. Typical values are: 0.12 µg/mL, which gives an impinger LOQ of 0.72 µg/cigarette and a pad LOQ of 0.96 µg/cigarette.
- Method Detection Limit (MDL)
- Stability of Reagents and Samples
- All primary stock and working KCN standards are prepared fresh weekly.
- All autoanalyzer reagents are prepared fresh weekly or as needed.
- All samples are analyzed within 24 hours of sample generation.
16 Modifications for Intense Smoking Conditions
- Under intense smoking conditions the number of cigarettes can be reduced from five to one or two.
17 Reference
- Collins, P.F. et al. A Trapping System for the Combined Determination of Total HCN and Total Gas Phase Aldehydes in Cigarette Smoke, Beitrage zur Tabakforschung, Vol 7, No.2, 1973.
- Rickert, W. S., and P. B. Stockwell. Automated determination of hydrogen cyanide, acrolein, and total aldehydes in the gas phase of tobacco smoke, J. Autom. Chem., 1, 1979, p. 152-154.
Appendix
Appendix 1: Graph of Colorimeter Output
The following contains a graph of colorimeter output generated by continuous flow colorimetric analyzer for various standards and samples.