Determination of Nitrate from Whole Tobacco
Health Canada
T-308 December 31, 1999
Table of Contents
- Scope of Applications
- Normative References
- Method Summary
- Apparatus and Equipment
- Reagents and Supplies
- Preparation of Glassware
- Preparation of Solution
- Preparation of Standards
- Sampling
- Sample Analysis
- Quality Control
- References
1 Scope of Applications
- This method describes the extraction of nitrate-nitrogen in whole tobacco and its determination using an automated continuous flow analyzer.
- This method is applicable to processed cigarette tobacco, finecut tobacco, chewing tobacco, cigar samples and pipe tobacco.
- This method is to be used to determine the amount of Nitrate added to whole tobacco as an antimicrobial agent. The method is designed to be used as a routine analysis without the need for extensive sample preparation or derivitization.
- This method does not distinguish between the amount of Nitrate added and the amount of naturally occurring Nitrate (if any) found in whole tobacco.
- This method also does not distinguish between Nitrate and Nitrite found in tobacco since all Nitrate is converted to Nitrite in the analysis.
2 Normative References
- American Society for testing and Materials (ASTM) D 1193-77 - Standard Specification for Reagent Water, Version 1977.
- Health Canada Test Method T-402 - Preparation of Cigarettes, Cigarette Tobacco, Cigars, Kreteks, Bidis, Packaged Leaf, Pipe and Smokeless Tobaccos for Testing, 1999-12-31.
- Health Canada Test Method T-115 - Determination of Tar, Water, Nicotine and Carbon Monoxide in Mainstream Tobacco Smoke, 1999-12-31.
3 Method Summary
- Ground whole tobacco is extracted with 5 % acetic acid and then filtered.
- The extract is then analyzed by an automated continuous flow colorimetric analyzer, where each sample undergoes on-line dialysis to remove interferences.
- The nitrate ion is reduced upon reaction with hydrazine at 37 °C in the presence of copper at a pH of 10.2.
- The nitrite formed is reacted with sulfanilamide under acidic conditions to yield a diazo compound which when mixed with N- (1-Napthyl)-ethylenediamine forms a red coloured complex.
- A single channel colorimeter with a 550 nm filter monitors the eluant. Nitrate found in whole tobacco is quantified by comparison to an external standard calibration.
Note: The testing and evaluation of certain products against this test method may require the use of materials and or equipment that could potentially be hazardous and this document does not purport to address all the safety aspects associated with its use. Anyone using this test method has the responsibility to consult with the appropriate authorities and to establish health and safety practices in conjunction with any existing applicable regulatory requirements prior to its use.
4 Apparatus and Equipment
- Robot Coupe Model RSI 2V Scientific Batch Processor or equivalent for grinding of tobacco.
- Analytical Balance capable of measuring to four decimal places.
- Clean 125 mL Polymethylpentene (PMP) Erlenmeyer flasks with screw-caps.
- Wrist-action shaker.
- 5 cc Disposable Syringe.
- Syringe Filter 0.45 µm PVDF (or equivalent).
- Computer Controlled Continuous Flow AutoAnalyzer (or equivalent) consisting of:
- Autosampler.
- Peristaltic Pump.
- Nitrate Manifold designed for AutoAnalyzer used.
- Single Channel Colorimeter equipped with 15 mm flow cell and 550 nm filter.
- 100 mL volumetric flasks with ground glass joints.
- 50 mL graduated cylinders.
- Glass filter funnel.
- Magnetic Stirrer.
- 1000 µL variable volume pipettor or equivalent (for standards preparation).
- Sample cups for autoanalyser.
- Disposable gloves.
- Protective eye glasses.
- 1 L volumetric flask.
5 Reagents and Supplies
- Acetic Acid, Glacial - HPLC Grade.
- Type I Water - as specified in ASTM D 1193).
- Hydrochloric Acid (HCl).
- ortho Phosphoric Acid (H3PO4), 98 %.
- Potassium Nitrate (KNO3).
- Cupric Sulfate Pentahydrate (CuSO4.5H2O).
- Methanol.
- Brij-35.
- Triton X-100.
- Hydrazine Sulfate (N2H4.H2SO4).
- Sodium Hydroxide (NaOH).
- Sodium Acetate Anhydrous (NaC2H3O2).
- Sulfanilamide(C6H8N2O2S).
- Sodium Nitrite (NaNO2).
- N-1-Napthylethylenediamine dihydrochloride (C12H14N2.2HCl).
6 Preparation of Glassware
- Glassware should be cleaned and dried in such a manner to ensure that contamination from glassware does not occur.
7 Preparation of Solution
Note: The large volumes of solution described in these preparations may not be necessary if only small volumes of sample are to be analyzed. It is important, however, to maintain the proper concentrations of solutions prepared.
- 50 % Triton X-100 Solution
- Mix 50 mL Triton X-100 with 50 mL methanol. Transfer to storage bottle.
- Colour Reagent
- Add 700 mL of Type I water to a 1 L volumetric flask.
- Add 100 mL of concentrated phosphoric acid.
- Dissolve 40 g of sulfanilamide in the above solution. Heat if necessary to dissolve completely.
- Add 2.0 g of N-1-Napthylethylenediamine dihydrochloride.
- Allow mixture to cool to room temperature, add 0.5 ml Brij-35 and make up to volume with Type I water.
- Store refrigerated (4 °C) in an amber bottle. The solution is stable for one month.
- Copper Sulfate Solution
- Cupric sulfate stock solution:
- Dissolve 1.25 g of cupric sulfate in Type I water and make to 500 mL.
- Add 4.0 mL of the cupric sulfate stock solution to Type I water in a 1 L volumetric flask.
- Add 0.5 mL Brij-35 and make up to volume with Type I water.
- Hydrazine Sulfate Solution
- Hydrazine sulfate stock solution
- Dissolve 25.0 g of hydrazine sulfate in 800 mL of Type I water. Dilute to 1 L. The stock solution is stable for six months if kept in a tightly stoppered amber bottle.
- Add 36 mL of stock hydrazine sulfate to 900 mL of Type I water in a 1 L volumetric flask.
- Add 0.5 mL Brij-35 and make up to volume with Type I water.
- Store in an amber bottle. Stable for one month.
- Hydrazine sulfate stock solution
- 0.2 N Sodium Hydroxide
- Dissolve 8.0 g of sodium hydroxide in about 800 mL of Type I water.
- Add 0.5 mL Brij-35, allow solution to cool and make up to 1 L with Type I water.
- Acetate Buffer (pH 3.7)
- Dissolve 16.4 g of anhydrous sodium acetate in about 300 mL of Type I water.
- Add 100 mL of acetic acid, 1 mL of 50 % Triton X-100 solution and dilute to 1 L.
- Dialyzer Solution
- Dilute 1 mL of Brij-35 to 1000 mL with Type I water.
- Tobacco Extraction Solution (5 % Acetic Acid)
- Dilute 50 mL of Glacial Acetic Acid to 1000 mL with Type I water.
- 1.0N Hydrochloric Acid
- Carefully add 83 mL hydrochloric acid to about 800 mL of Type I water in a 1 L volumetric flask. Mix well.
- Cool and make up to volume with Type I water.
- This solution is used for the cleaning of the Copper Sulfate lines of the AutoAnalyzer to prevent the build up of copper between projects or groupings of analyses.
8 Preparation of Standards
- Primary Standard (1°) Potassium Nitrate (2000ppm Nitrogen)
- Dissolve 14.442 g of dessicated potassium nitrate in about 800 mL of 5 % acetic acid extraction solution (in a 1 L volumetric flask)
- Dilute to volume with 5 % acetic acid extraction solution.
- Potassium Nitrate Calibration Standards
- Take appropriate volumes (0.20 to 3.0 mL) of the nitrate primary stock solution and dilute to 100 mL with the extraction solution to give calibration standards with approximate nitrogen (from nitrate) concentrations in the ranges detailed below.
The following table displays the details for the preparation of potassium nitrate calibration standards. The table includes the volume of the primary KNO3 standard, final volume of the calibration standard, and the concentration of nitrogen in parts per million in the final calibration standard.
Standard ID Vol (mL) 1°KNO3 Std Final Volume in mL Nitrogen[ppm] Blank 0 0 100 0.0 Std 1 0.2 100 4 Std 2 0.5 100 10 Std 3 1.0 100 20 Std 4 2.0 100 40 Std 5 3.0 100 60 - Nitrate calibration standards are prepared fresh every five working days.
- Take appropriate volumes (0.20 to 3.0 mL) of the nitrate primary stock solution and dilute to 100 mL with the extraction solution to give calibration standards with approximate nitrogen (from nitrate) concentrations in the ranges detailed below.
- Primary Stock (1°) Sodium Nitrite (2000 Nitrogen)
- Dissolve 9.852 g of dessicated sodium nitrite in about 800 mL of 5 % acetic acid extraction solution (in a 1 L volumetric flask).
- Dilute to volume with 5 % acetic acid extraction solution.
- Sodium Nitrite Calibration Check Standard
- Take 2.0 mL of the nitrite primary stock solution and dilute to 100 mL with the extraction solution to give a 40 ppm nitrogen (from nitrite) calibration standard.
- Use immediately to check the reduction efficiency.
- Nitrite calibration standards are prepared fresh as required.
9 Sampling
- The sampling of tobacco products for the purpose of testing shall be as specified in T-115.
10 Sample Analysis
- Preparation of Test Sample
- The whole tobacco required for analysis must be removed from its original package and inspected for extraneous material.
- Prepare product as per Health Canada Test Method T-402.
- The product may be analyzed on an "as received" basis and the results corrected to a "dry matter" basis, if required, by the determination of moisture on the homogenized product.
- Extraction of Whole Tobacco
- Accurately weigh 0.5 g of homogenized or ground tobacco into a 125 mL PMP Erlenmeyer flask with cap.
- Add 50 mL of 5 % acetic acid to the flask and cap.
- With each group of whole tobacco samples tested:
- Prepare a laboratory reagent blank (LRB) as follows to demonstrate that interferences from the analytical system, glassware, and reagents are not present.
- LRB: Add 50 mL of 5 % extraction solution to an Erlenmeyer flask and cap.
- Prepare an LFB as follows to determine whether there is any loss of analyte as a result of the analytical process.
- LFB: Add 1mL of the 1° Potassium Nitrate Stock to a clean Erlenmeyer flask, add 49 mL of the 5 % acetic acid extraction solution and cap.
- Clamp flasks onto armature of the wrist action shaker and agitate 15 minutes.
- The whole tobacco extract is then filtered directly into appropriately labelled autoanalyzer sample cups using a syringe filter attached to a 5 cc disposable syringe.
- After shaking, prepare an laboratory fortified matrix (LFM) using a standard control brand as follows:
- LFMA: Preload a volumetric flask with 5 mL of 5 % acetic acid extraction solution. Attach a 0.45 µm PVDF filter to a 5 cc disposable syringe and filter the tobacco extract directly into 10 ml volumetric flask, make up to the mark.
- LFMB: Preload a volumetric flask with 4.9mL of 5 % acetic acid extraction solution plus 0.1 mL of the 1° Potassium Nitrate Stock solution. Attach a 0.45 µm PVDF filter to a 5 cc disposable syringe and filter the tobacco extract directly into 10 mL volumetric flask up to the mark.
- Instrument Analysis
- Continuous Flow Analyzer setup
- Refer to diagram below (Diagram 1).
- This setup is specific to a Technicon AutoAnalyzer system. The use of a different manufacturer's system may require different flows and/or components.
The following figure is a schematic flow diagram of a technicon auto analyzer, including the detector and heating bath connected on the left of the primary dialyzer, and the sampler on the right of the dialyzer.
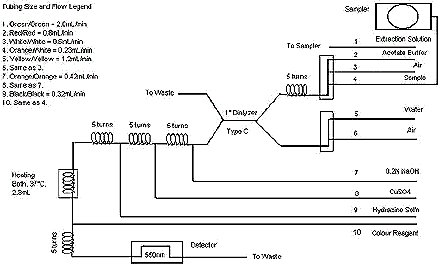
Diagram 1: Typical Flow Diagram for Technicon AutoAnalyzer
- Reduction Efficiency Check
- Each working hydrazine solution must be checked for efficient reduction capacity. Run both the 40 ppm nitrate and the nitrite standards. If the apparent nitrogen from nitrate concentration (response) is much lower than the nitrogen from nitrite concentration, adjust the working hydrazine solution by adding 1 mL aliquots of the hydrazine stock solution until the two standards show equal response.
- Continuous Flow Analysis
- The Autosampler is operated in the normal manner at a sampling rate of 20 per hour with a 2:1 sample to wash ratio. Sufficient time should be allowed for the system to become stable with the reagents being pumped. Conditions may vary depending on the AutoAnalyzer being used.
- Sample vials are loaded onto the Autosampler such that calibration standards are first in the queue, followed by a baseline correction, then the tobacco extract samples such that every 10th vial is a standard (run as a sample) and then finally a baseline correction at the end.
- Samples are rerun only if the analyte response is out of range or there was a particular problem with the analysis.
- Samples undergo on-line dialysis.
- Sampling cups containing only 5 % acetic acid (extraction solution) are placed at regular intervals to allow for baseline correction.
- The 5 % extraction solution is also used as the sample wash.
- Continuous Flow Analyzer setup
- Calculations
- Construct a Calibration Curve:
- A calibration curve for nitrogen is prepared by plotting the concentration of the standards versus their respective peak heights, in order to determine a response factor (RF).
- Sample Quantification
- The amount of nitrogen in the extracted solutions is quantified by an external standard method, by multiplying the height response of the solution by the response factor (RF).
Nitrogen (µg/mL) = sample height X RF.
- The amount of nitrogen in the extracted solutions is quantified by an external standard method, by multiplying the height response of the solution by the response factor (RF).
- Determination of Nitrogen (µg/g) from Nitrate
- By entering the correct multiplier (overall volume the original sample is diluted to (50 mL)) and divisor (the original sample weight 0.5 g) the concentration of nitrogen from nitrate is automatically calculated in µg/g:
Nitrogen (µg/g) = [ppm nitrogen (µg/mL) x 50 (mL)] / 0.5 g sample
- By entering the correct multiplier (overall volume the original sample is diluted to (50 mL)) and divisor (the original sample weight 0.5 g) the concentration of nitrogen from nitrate is automatically calculated in µg/g:
- Percent Nitrogen may be calculated using the following:
Nitrogen (%) = ppm nitrogen (µg/g) / 10000 - All results are expressed on an "as received" basis. These may be expressed on a dry matter basis using the appropriate moisture result.
- All results may be reported as Nitrate (%) or Nitrate (µg/g) by multiplying the Nitrogen results by 4.43 (the relative weight of Nitrate to Nitrogen).
Nitrate (µg/g) = Nitrogen (µg/g) X 4.43.
Nitrate (%) = Nitrogen (%) X 4.43.
- Construct a Calibration Curve:
11 Quality Control
- Recoveries and Levels of Contamination
- Each analytical run should also include:
- A Laboratory Reagent Blank (LRB) to evaluate the extent of any interferences due to glassware, reagents, and analyzer effects. Typical LRB results are ND (Not Detected).
- A Laboratory Fortified Blank (LFB) to evaluate the extent of potential analyte loss.
- A Laboratory Fortified Matrix (LFM) to assess matrix interferences. This is accomplished by spiking a true sample with a known concentration and determining a per cent recovery.
- Each analytical run should also include:
- Method Detection Limit (MDL) and Limit of Quantitation
- Method Detection Limit (MDL)
The method detection limit is determined by analyzing the lowest level standard at least 10 times as an unknown over several days. The MDL is then calculated as three times the standard deviation of these determinations. - Limit of Quantitation (LOQ)
The limit of quantitation is determined by analyzing the lowest level standard at least 10 times as an unknown over several days. The LOQ is then calculated as 10 times the standard deviation of these determinations.
- Method Detection Limit (MDL)
- Stability of Reagents and Supplies
- Stock Hydrazine Sulphate is stable for up to six months when stored at room temperature (±20 °C) in a tightly stoppered amber bottle.
- Hydrazine Sulphate Working Solution is stable for up to one month when stored at room temperature (±20 °C).
- Sodium Nitrite stock and working standard must be prepared as required due to instability of the solutions.
- The Sulfanilamide Colour Reagent is stable for up to one month when stored at 4 °C in an amber bottle.
- All primary stock Nitrate standards are prepared fresh weekly.
- All Nitrate working standards, and extraction solvents are prepared fresh weekly.
- All samples are to be analyzed within 24 hours.
12 References
- AOAC Official Method 968.07, Nitrogen (Nitrate and Nitrite) in Animal Feed, Colorimetric Method, AOAC Official Methods of Analysis (1995), Chapter 4, p. 14 -15.
- Industrial Method No. 838 - 87T; Nitrate in Tobacco Extracts, Bran + Leubbe Inc., Technicon Industrial Systems Corp., 1025 Busch Parkway, Buffalo Grove, Illinois, USA 60090 - 4516.