Determination of Triacetin in Whole Tobacco
Health Canada
T-311 December 31, 1999
Table of Contents
- Scope of Applications
- Normative References
- Method Summary
- Apparatus and Equipment
- Reagents and Supplies
- Preparation of Glassware
- Preparation of Solutions
- Preparation of Standards
- Tobacco Product Preparation
- Smoking Machine Preparation
- Sample Generation
- Sample Analysis
- Quality Control
- References
- Appendices
1 Scope of Applications
- The flavour and aroma of tobacco and tobacco smoke are important qualities to tobacco users and the use of additives in the blending of pipe, snuff and chewing tobacco has long been a common practice. This method is used to determine the amount of Triacetin added to whole tobacco as a flavorant by acidic steam distillation and its quantification by gas chromatography with flame ionization detection.
- The method is designed to be used as a routine analysis without the need for derivitization. The expected amount is in the range of 14.9 mg per 20 mm filter tip and no triacetin is found in cigarettes without filter tips. This is applicable to processed cigarette tobacco, finecut tobacco, pipe, snuff, chewing tobacco and cigars.
2 Normative References
- American Society for Testing and Materials (ASTM) D 1193-77 - Standard Specification for Reagent Water, Version 1977.
- Health Canada Test Method T-115 - Determination of Tar, Water, Nicotine and Carbon Monoxide in Mainstream Tobacco Smoke, 1999-12-31.
3 Method Summary
- Whole tobacco is steam distilled in 0.5 N Sulphuric Acid.
- The steam distillate is extracted three times with methylene chloride.
- The combined extract is then rotary-evaporated to near dryness and concentrated under nitrogen.
- The sample is then analyzed by gas chromatography using a split mode of injection with a flame ionization detector (FID).
- Quantification is achieved by external standard calibration procedures where the relative response of the samples is compared against a five-point calibration.
Note: The testing and evaluation of certain products against this test method may require the use of materials and or equipment that could potentially be hazardous and this document does not purport to address all the safety aspects associated with its use. Anyone using this test method has the responsibility to consult with the appropriate authorities and to establish health and safety practices in conjunction with any existing applicable regulatory requirements prior to its use.
4 Apparatus and Equipment
- 250 mL Erlenmeyer flasks.
- 2000 mL volumetric flasks.
- 100 mL volumetric flasks.
- 10 mL volumetric flasks.
- 500 mL separatory funnels.
- 250 mL graduated cylinder.
- 250 mL distillation flasks.
- Steam Distillation Unit.
- Rotary Evaporator (Rotovap or equivalent).
- Batch Processor for grinding of tobacco (Robot Coupe RS1-2V or equivalent).
- Micropipettes for the preparation of analytical standards (0.050 mL, 0.200 mL and 0.500 mL) (or a variable pipettor optional).
- Zymark TurboVap Evaporator or equivalent.
- Pipettes - Class A 2.0 mL and 8.0 mL.
- 250 mL round bottom flasks with ground glass joints.
- PC-controlled Gas Chromatograph with:
- Split / Splitless Injector.
- Autosampler.
- Flame Ionization Detector.
- Work Station.
- OV101 fused silica capillary column.
- Analytical Balance capable of reading to four decimal places.
- Disposable gloves.
- Protective eye glasses.
- Autosampler Vials with screw caps.
- 15 mL graduated Conical centrifuge tubes.
5 Reagents and Supplies
Note: All reagents shall be, at the least, recognized as reagent grade in quality.
- Triacetin (liquid) > 99 % purity.
- Methylene Chloride - Distilled in glass (DIG).
- Methanol - DIG.
- Sulphuric Acid, 95 - 97 %.
- Type I water as specified in ASTM D1193.
- Ultra High Purity (UHP) Helium (Carrier Gas).
- UHP Hydrogen (FID).
- Extra - Dry Air (FID).
- UHP Nitrogen (Makeup).
- HP Nitrogen (TurboVap).
- Parafilm® or equivalent.
- Septa, 8mm Teflon-faced, 60 mil.
- Disposable Glass Pasteur Pipettes.
- Rubber Bulbs.
- Glass Beads.
6 Preparation of Glassware
- Glassware should be cleaned and dried in such a manner to ensure that contamination from glassware does not occur.
7 Preparation of Solutions
- Not applicable to this method.
8 Preparation of Standards
- Primary Stock (1°) Triacetin Standards
- Weigh 100 mg of the liquid Triacetin into a 100 mL volumetric flask.
- Dilute to volume with methanol.
- Triacetin Calibration Standards
- Take appropriate volumes (0.050 to 8.0mL) of the triacetin primary stock solution and dilute to 10mL with methylene chloride to give calibration standards with approximate triacetin concentrations in the ranges detailed below.
The following table contains triacetin calibration standards including sample Blank 0 and standards 1 to 5. The table also includes the volume of primary standard, the final volume, and the concentration of triacetin.
Triacetin Calibration Standards Standard
IDVolume (mL) 1°
Triacetin StdFinal Volume
in mLTriacetin
[μg/mL]Blk 0 0 10 0.0 Std 1 0.050 10 5 Std 2 0.200 10 20 Std 3 0.500 10 50 Std 4 2.0 10 200 Std 5 8.0 10 800 Note 1: The concentration of Triacetin will vary depending on the exact concentration of primary stock prepared. - Transfer to 1.5 mL Varian amber autosampler vials. Rinse vial first and then fill to minimize headspace.
- Place vials in a vial file and store at 4 °C, protected from light, until instrument analysis is performed.
- Triacetin calibration standards are prepared fresh every five working days.
- Take appropriate volumes (0.050 to 8.0mL) of the triacetin primary stock solution and dilute to 10mL with methylene chloride to give calibration standards with approximate triacetin concentrations in the ranges detailed below.
9 Tobacco Product Preparation
- Preparation of Test Sample
- The whole tobacco required for analysis must be removed from its original package and inspected for extraneous material.
- Whole tobacco samples are to be finely chopped in the Robot Coupe processor to a 20 mesh particle size.
- Ground whole tobacco samples are conditioned as specified in T-115.
10 Smoking Machine Preparation
- Not applicable to this method.
11 Sample Generation
- Accurately weigh 2 g of conditioned, ground whole tobacco into a 250 mL distillation flask.
- Add 150 mL of the aqueous 0.5 N sulphuric acid solution and a few glass beads to the 250 mL distillation flask.
- Add approximately 25 mL of Type I water to the bottom of another 250 mL flask placed at the condenser outlet end of the steam distillation unit. The end of the outlet tube must be immersed in the water.
- Start the steam distillation unit and collect approximately 200 mL of distillate.
- Record the flow rate of tap water through steam distillation, for consistency and to ensure optimal recovery of triacetin.
- Transfer the distillate to a 500 mL separatory funnel and add 50 mL of methylene chloride.
- Shake the separatory funnel and then drain off the methylene chloride. Transfer the extracted layer to a 250 mL round-bottomed flask.
- Repeat the extraction procedure a total of three times.
- Combine the three extracts in a round-bottomed flask.
- Evaporate the sample at room temperature on a rotary evaporator to approximately 5 mL.
- Quantitatively transfer the residual to a 15 mL graduated conical centrifuge tube with 2 × 2 mL rinses of methylene chloride.
- Evaporate the sample under a constant stream of nitrogen on the TurboVap to 1 mL.
- Using a disposable Pasteur pipette, transfer the sample to a 1.5 mL amber autosampler vial.
- Place the vial in a vial file and store at 4 °C, protected from light, until instrument analysis is performed.
- Prepare an LRB, LFB and LFM (see section 13) with each run of whole tobacco samples as follows to demonstrate that interference from the analytical system, glassware, and reagents are not present.
- LRB: Steam distill 150 mL of 0.5 N sulphuric acid and treat as a sample.
- LFB: Add 0.1 mL of the triacetin primary stock and 140 mL of 0.5 N sulphuric acid to a distillation flask. Treat as a sample.
- LFM: Add 0.1 mL of the triacetin primary stock to a duplicate sample of whole tobacco. Treat as a sample.
12 Sample Analysis
- Gas Chromatograph Configuration
- Injector: Splitless mode with (20:1) ratio
- Silanized Insert (Splitless)
- Column: HP - 101 fused silica capillary column
- Detector: Flame Ionization (FID)
- Channel A, 1 Volt Full Scale.
- Range: 12 Atten: 8.
- Autozero: On.
- Carrier: He at 40.0 psi, flow = 2.3 mL/minute @ 70 °C.
- Split (purge flow) = 50 mL/minute.
- Relays: Initial relay state: On.
- Relay time: 0.40 minutes to Off.
Note: Detector gas flows set as per manufacturer's specification. - Gas Chromatograph Operating Conditions
- Injector: 220 °C.
- Detector: 250 °C.
- Column Oven Temperature Profile:
Start Temperature: 70 °C Hold for 2.0 minutes.
Rate: 5.0 °C/minute to 220 °C Hold for 3.0 minutes.
Total Run Time: 35.00 minutes.
- Autosampler Conditions
- Injection volume: 1.0 µL.
- Solvent Select: Hexane; then Methanol.
- Solvent flush sampling: Yes.
- Solvent plug: 0.1 µL.
- Upper Air Gap: Yes.
- Lower Air Gap: Yes.
- Syringe wash time: 20 seconds.
- Uptake speed: 1.0 µL/second.
- Injection rate: 10.0 µL/second.
- Hot needle time: 0.02 minutes.
- Needle resonance time: 0.01 minutes.
- Pause time: 2.0 seconds.
- Analysis
- Sample vials are loaded onto the autosampler such that every 10th vial is a standard solution and in such quantities that the total time (sample generation plus analysis time) does not exceed 24 hours at room temperature.
- One µL of each sample vial is injected onto the GC column and analyzed.
- Calculations
- Construct a Calibration Curve:
- One µL of each calibration standard is injected onto the GC column and analyzed as per the chromatographic conditions specified.
- A calibration curve of triacetin is prepared by plotting the concentration of the standards versus their respective peak areas.
- Determine the response factor from the calibration curve.
- Sample Quantification
- The amount of the triacetin flavourant in whole tobacco samples is quantified by the external standard method. See Figure 3, Overlay of Chromatograms.
- The identification of peaks is by comparison of retention times with standards, and the spiking of samples.
- Construct a Calibration Curve:
- Determination of Triacetin Deliveries in [µg/cigarette]
Triacetin [µg/g] = [Peak Area-Int / Resp. Factor] × [mL of solution / Wt of Tobacco]
- By entering the correct multiplier (overall volume the original sample is diluted to in mL) and divisor (the original sample weight in g) the concentration of triacetin is automatically calculated in µg/g.
- To convert this concentration to a percentage (%), the µg/g result must be divided by 10000.
- All results are expressed on an "as conditioned" basis. These may be expressed on a dry matter basis using the appropriate moisture result.
13 Quality Control
- Typical Chromatograms
- See Figures 1, 2 and 3.
- Recoveries and Levels of Contamination
- Each analytical run of test cigarettes should also include:
- A Laboratory Reagent Blank (LRB) to evaluate the extent of any interference due to glassware, reagents and analyzer effects.
- A Laboratory Fortified Blank (LFB) to evaluate the extent of potential analyte loss.
- A Laboratory Fortified Matrix (LFM) to assess matrix interference. This is accomplished by spiking a true sample with a known concentration and determining a per cent recovery.
- Each analytical run of test cigarettes should also include:
- Method Detection Limit (MDL) and Limit of Quantitation
- Method Detection Limit (MDL)
- The method detection limit is determined by analyzing the lowest level standard at least 10 times as an unknown over several days. The MDL is then calculated as three times the standard deviation of these determinations.
- Limit of Quantitation (LOQ)
- The limit of quantification is determined by analyzing the lowest level standard at least 10 times as an unknown over several days. The LOQ is then calculated as 10 times the standard deviation of these determinations.
- Method Detection Limit (MDL)
- Stability of Reagents and Supplies
- All primary stock Triacetin standards are prepared fresh weekly.
- All work standards, and extraction solvents are prepared fresh weekly.
- All samples are to be analyzed within 24 hours.
14 References
- LaVoie, E.J., Shigematsu, A., Tucciarone, P.L., Adams, J.D., and Hoffmann, D., 1985. Comparison of the Steam-Volatile Components of Commercial Cigarette, Pipe, and Chewing Tobaccos, Journal of Agricultural and Food Chemistry 33, p. 876-879.
Appendices
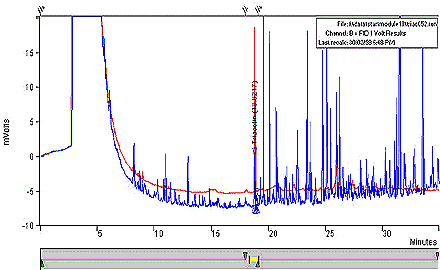
Figure 1: Chromatogram of a Typical Triacetin Calibration Standard
This figure is a chromatogram of a typical triacetin calibration standard. The voltage was measured in mVolts over a period of time, in minutes.
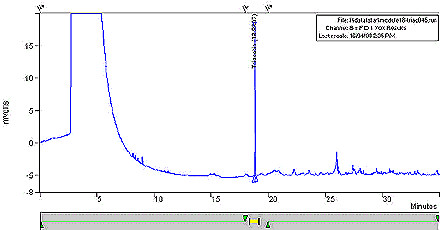
Figure 2: Chromatogram of the Analysis of Whole Tobacco (Control)
This figure is a chromatogram of the analysis of whole tobacco (control). The voltage was measured in mVolts over a period of time, in minutes.
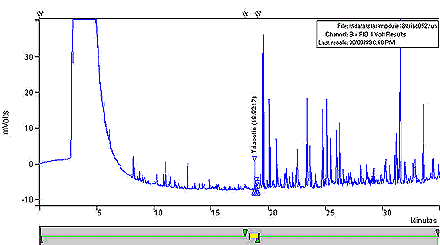
Figure 3: Overlay Chromatograms of Control Whole Tobacco and Triacetin Calibration Standard
This figure is an overlay of chromatograms of control whole tobacco and triacetin calibration standard.