Determination of Phenolic Compounds in Sidestream Tobacco Smoke
Health Canada
T-211, December 31, 1999
Table of Contents
- Scope of Applications
- Normative references
- Definitions
- Method Summary
- Apparatus and Equipment
- Reagents and Supplies
- Preparation of Glassware
- Preparation of Solutions
- Preparation of Standards
- Sampling
- Tobacco Product Preparation
- Smoking Machine Preparation
- Sample Generation
- Sample Analysis
- Quality Control
- Modifications for Intense Smoking Conditions
- Reference
- Appendices
1 Scope of Applications
- This method describes the extraction and determination of phenolic compounds in the sidestream (SS) tobacco smoke by reversed phase high performance liquid chromatography (HPLC).
- Applicable to the trapping and quantitation of phenolic compounds in sidestream tobacco smoke on the sidestream glass fibre filter disc (pad) and impinger only.
2 Normative References
- American Society for Testing and Materials (ASTM) D 1193-77 - Standard Specification for Reagent Water, Version 1977.
- Health Canada Test Method: T-115 - Determination of Tar, Water, Nicotine and Carbon Monoxide in Mainstream Tobacco Smoke, 1999-12-31.
3 Definitions
- Refer to T-115 for definitions of terms used in this document.
4 Method Summary
- 10 equidistant ports of a standard 20 port linear smoking machine are reconfigured with the BAT (British American Tobacco) fishtail chambers and flow-controlled vacuum pumps.
- Cigarettes or other tobacco products are smoked beneath the fishtail chambers and the smoke is swept up the chimney at the rate of 3 L/minute.
- The total particulate matter (TPM) from the sidestream smoke is collected on a pad at the top of the chimney. The filtered puff is then bubbled through an impinger containing 100 mL of 1 % acetic acid.
- After smoking two cigarettes*, the sidestream pad is placed in a glass-stoppered Erlenmeyer flask that contains the impinger solution and 2 X 20 mL rinses of the BAT fishtail chamber and is extracted by wrist-action shaking.
*For other tobacco products, select a number such that breakthrough does not occur. - An aliquot of the TPM extract is then syringe filtered and subjected to reversed-phase gradient high performance liquid chromatography (HPLC).
- Phenols are monitored using selective fluorescence detection and quantified by comparison to an external standard calibration.
- The sample generation and analysis should be completed in one day.
Note: The testing and evaluation of certain products against this test method may require the use of materials and or equipment that could potentially be hazardous and this document does not purport to address all the safety aspects associated with its use. Anyone using this test method has the responsibility to consult with the appropriate authorities and to establish health and safety practices in conjunction with any existing applicable regulatory requirements prior to its use.
5 Apparatus and Equipment
- Equipment needed to perform conditioning as specified in T-115.
- Equipment needed to perform marking for butt length as specified in T-115.
- Equipment needed to perform smoking of tobacco products as specified in T-115.
- Glass fibre filter holders.
- Glass fibre filter discs (pads), 44 mm in diameter, with no more than 5 % acrylic type binder.
- Analytical Balance capable of measuring to at least four decimal places.
- Wrist Action Shaker.
- Vacuum Pumps.
- Flow meters.
- Fishtail Chambers - BAT (10).
- Glass Impingers with frits and cooling jackets - 10 X 10", capacity 250 mL.
- Cooling Bath.
- 250 mL Erlenmeyer flasks with ground glass stoppers.
- Volumetric flasks 10 mL, 25 mL and 50 mL, Actinic Red.
- Glass Micropipettes - assorted volumes (100, 150, 300, 400, 500, 800, 1000, and 2000 μL).
- Glass Transfer Pipettes - 1, 2, 5, 6, 7, 8, and 20 mL.
- Glass Graduated Measuring Cylinders 25 mL and 50 mL.
- Erlenmeyer flasks with ground glass joints 50 mL, Actinic Red.
- High Pressure Liquid Chromatography System consisting of:
- Solvent Delivery System - tertiary gradient pump.
- Refrigerated Autosampler with 20 μL sampling loop.
- Programmable Wavelength Spectrofluorometer.
- Column Temperature Modifier.
- Cooling Bath.
- Data collection system.
- RP18e 250 mm X 4 mm and 5 μm Column with 10 mm X 4 mm guard column.
6 Reagents and Supplies
Note: All reagents shall be, at the least, recognized as analytical reagent grade quality.
- Syringe Filters 0.45 μm PVDF.
- Disposable syringes.
- Disposable Glass Pasteur Pipettes.
- Rubber Bulbs.
- Autosampler vials, screw caps and septa.
- Masking Tape.
- Aluminum Foil.
- Methanol - Distilled in Glass (DIG).
- Acetonitrile - DIG.
- Isopropanol (IPA) - DIG.
- Ethanol - DIG.
- Acetic Acid - HPLC grade.
- Octanol > 99 % purity.
- Type I water (meets ASTM D 1193 specification).
- Hydroquinone > 99 % purity.
- Resorcinol > 99 % purity.
- Catechol > 99 % purity.
- Phenol > 99 % purity.
- m-Cresol > 99 % purity.
- p-Cresol > 99 % purity.
- o-Cresol > 99 % purity.
7 Preparation of Glassware
- Glassware should be cleaned and dried in such a manner to ensure that contamination from glassware does not occur.
8 Preparation of Solutions
- Prepare 4 L fresh 1 % acetic acid solution in Type I water (40 mL diluted up to 4 L) and test by HPLC for contamination.
9 Preparation of Standards
- Primary (1°) Phenol Standards (See Appendix 1)
- Weigh 25 mg of the following phenols (Hydroquinone, Resorcinol, Catechol, Phenol, m-Cresol, p-Cresol and o-Cresol) into individual 25 mL volumetric flasks and make up to the mark with fresh 1 % acetic acid solution.
- Concentrations will be in the range of 1.0 mg/mL. Prepare fresh primary phenol stock standards every 10 working days.
- Secondary (2°) Phenol Standard Solutions (See Appendix 1)
- Take appropriate volumes of the 1° Phenol Standards and dilute to 10 mL with 1 % acetic acid.
- Prepare 2° phenol stock standards fresh with each new primary stock standards.
- Tertiary (3°) Phenol Solution (See Appendix 1)
- Take corresponding volumes of each phenol solution and add to a single 50 mL volumetric flask. Dilute up to the mark with 1 % acetic acid.
- Prepare phenol working stock solution fresh every five working days.
- Phenol Working Standards
- Take appropriate volumes (0.100 to 7.5 mL) of the Tertiary (3°) Stock Phenol solution and dilute to 10 mL with 1 % acetic acid to give calibration standards with appropriate phenol concentrations.
- Transfer to autosampler vials.
- Phenol calibration standards are prepared fresh every five working days.
- Phenol Spiking Solution for laboratory fortified blanks (LFB)
- Add selected volumes of the phenol stock standards together in a 50 mL volumetric flask and make up to the mark with 1 % acetic acid.
- Prepare phenol spiking solution fresh every five working days.
10 Sampling
- The sampling of tobacco products for the purpose of testing shall be as specified in T-115.
11 Tobacco Product Preparation
- Product shall be conditioned as specified in T-115.
- Cigarettes, cigarette equivalents, bidis, kreteks and cigars shall be marked for butt length as specified in T-115.
- Cigarettes to be smoked under intense smoking conditions shall be prepared as specified in T-115.
12 Smoking Machine Preparation
- Ambient Conditions
- The ambient conditions for smoking shall be as those specified in T-115.
- Machine Conditions
- The machine conditions shall be as those specified in T-115 with the following modifications as detailed below:
- Assemble the Phenol sidestream apparatus as per the diagram.
- Raise chimney level to highest position (loading position).
- The sidestream impinger is loaded with 100 mL of 1 % acetic acid plus two drops of octanol and the impinger jackets are connected to a cooling bath at 10 °C.
- Install the weighed sidestream filter pad assembly at the top of the fishtail chamber and place impingers with tops connected to coarse frits onto the rear section of smoke machine.
- Tubing from impinger front (internal stem connection) attaches to SS filter pad holder and from impinger rear (bulb) to vacuum pump.
- Calibrate the vacuum pumps to draw at the rate of 3 L/minute. Record the flow meter settings.
- Attach the mainstream filter pad holders to the corresponding port on the smoking machine.
The following figure demonstrates sidestream apparatus. The BAT fishtail chimney contains a mainstream pad which connects to the port of the smoking machine at its base; whereas the tygon tube is connecting the fishtail to the sidestream pad holder on the top of the chimney. The sidestream pad holder on the BAT fishtail chimney is connected to the 250 mL impinger containing the extraction solution, by tygon tubing. The impinger is then connected outwards by plastic tube connectors to a flow meter, and is also further connected to a pump that controls the flow rate of the exhaust.
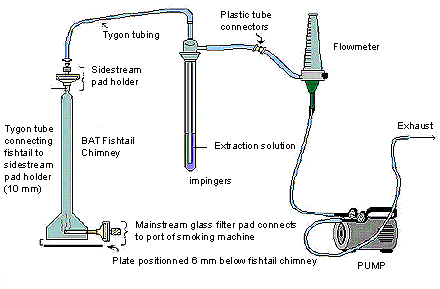
13 Sample Generation
Note: It is important to ensure that at least 50 to 100 mg of TPM has been deposited on the sidestream filter pad before proceeding with the analysis.
- Using the vacuum bar, install the first test cigarette to be smoked in position below the fishtail in the 10 calibrated ports. Gently insert the cigarette into the cigarette holder to the marked butt length.
- Turn on the sidestream pumps (3 L/minute) at the beginning of the lighting procedure at t minus 30 seconds.
- Light the cigarette (on the first puff) and initiate the puff count according to the following schedule:
- Normal lighting procedure is 15 second warm-up beginning at t-18 seconds followed by five seconds ignition. (Three seconds prior to puff plus the two seconds puff).
- Lower the fishtail assembly over the cigarette to a position of 6 mm above a plate that is beneath the cigarette. Do not allow the cigarette to touch the chimney. This is to create a uniform flow of air around the cigarette and up the fishtail chimney.
- The test cigarettes are smoked to the previously marked standard butt length. Extinguish and remove from beneath the BAT fishtail chamber.
- The pump continues for an additional 30 seconds to sweep any residual smoke up to the sidestream pad.
- The smoking process is repeated for the second cigarette.
- Smoking is terminated when the final test cigarette has been consumed to the predetermined end mark.
- At the end of the smoking process raise the chimney and disassemble the sidestream apparatus.
- Re-weigh and record the "after smoking" weights of the sidestream filter holders.
14 Sample Analysis
- Extraction of filter pads
- One run consists of 10 samples (pads). Process 10 samples at a time but not more than two runs or 20 samples per day. Do not smoke more than can be analyzed in a 24 hours period. Hydroquinone is especially temperature and time sensitive.
- Remove the sidestream pad, fold in half and in half again with the "clean" side facing out. Grasp with a pair of clean tweezers, and wipe the holder. Place the pad into a 250 mL Erlenmeyer flask.
- Add the 100 mL of impinger solution to the Erlenmeyer flask.
- Rinse the fishtail chimney with 2 X 20 mL of fresh 1 % Acetic Acid. Use a glass rod to free up any debris on the chimney. Add the chimney washings to the Erlenmeyer flask for a total volume of 140 mL.
- Place a piece of 1" masking tape over the ground glass stopper to hold it in place.
- Prepare an Laboratory Reagent Blank (LRB) with each day's smoking as follows to demonstrate that interference from the analytical system, glassware, and reagents are not present.
- LRB: Add one blank filter pad from the smoking room to a clean 250 mL Erlenmeyer flask, add 140 mL of 1 % Acetic Acid solution and stopper.
- Prepare an Laboratory Fortified Blank (LFB) with each day's smoking as follows to determine whether there is any loss of analyte as a result of the analytical process.
- LFB: Add one blank filter pad from the smoking room to a clean 250 mL Erlenmeyer flask, add 139 mL of 1 % acetic acid plus 1 mL of phenol spiking solution and stopper.
- Wrap flasks with tin foil completely. Clamp flasks onto armature of wrist action shaker and agitate 30 minutes. The pad should be disintegrated once agitation is complete.
- After shaking, syringe-filter the smoke extract directly into autosampler vials in duplicate. Rinse the vial first, discard the rinse; and then fill to minimize headspace.
- After shaking, prepare an Laboratory Fortified Matrix (LFM) using a standard control brand with each day of smoked samples:
- Attach a 0.45 μm syringe filter to a disposable syringe and filter the smoke extract directly into 10 mL volumetric flasks that have been preloaded with 1 % acetic acid and the phenol spike as necessary to dilute the smoke extract to 10 mL. Mix the volumetric flask well and then using a Pasteur pipette, fill autosampler vials in duplicate. Rinse vial first and then fill to minimize head space.
- The LRB and LFB are syringe-filtered directly into autosampler vials.
- Place the vials in a vial file and store at 4 °C, protected from light, until instrument analysis takes place.
- A run log is then generated to record the total time samples are at room temperature from smoking to the end of analysis.
Note: It is very critical that analysis be completed in minimal time without interruption as the samples will decompose with prolonged exposure at room temperature.
- Instrument Analysis: HPLC Equipment
- High Pressure Liquid Chromatography System consisting of:
- Solvent Delivery System - ternary gradient pump.
- Refrigerated Autosampler with 20 μL sampling loop.
- Programmable Wavelength Spectrofluorometer at Gain 100, ATTN 8.
- Slit Width: Ex. 18 nm, Em 18 nm.
- Wavelength Profile:
Time Initial Excitation (nm) Emission (nm) 0.0 304 338 5.5 274 298 32.0 274 298 33.5 304 338 - Cooling Bath with column temperature modifier attachment.
- Chromatographic Conditions (Reversed Phase Analysis)
- Column Temperature: 20 °C.
- Mobile Phase: Reagents.
- Solvent A: Prepare 2 L of 1 % Acetonitrile, 1 % Acetic Acid, 1 % IPA filter and degas. (UHP Helium sparged).
- Solvent B: Prepare 2 L of 28 % Acetonitrile, 1 % Acetic Acid, 1 % IPA filter and degas. (UHP Helium sparged).
- Solvent C: Acetonitrile (UHP Helium sparged).
- Sample Wash: Solvent A.
- Mobile Phase: Gradient.
Flowrate: 1.5 mL/minute.
Time (minutes) Composition % A % B % C 0.00 100 0 0 5.0 100 0 0 15.0 75 25 0 20.0 25 75 0 28.0 0 100 0 30.0 0 0 100 32.0 0 0 100 34.0 95 0 5 Method End Action: 100 0 0 (Equilibrate 10 minutes).
- Sample vials are loaded onto the autosampler such that every 10th vial is a standard solution and in such quantities that the total analysis time does not exceed 24 hours.
- Twenty μL of each sample vial is injected onto the HPLC. Elution pattern should be similar to Figure 1.
- Calculations
- Construct a Calibration Curve
- Twenty μL of each calibration standard is injected onto the HPLC column and analyzed as per the chromatographic conditions. Do in duplicate. Elution pattern should be similar to Figure 2.
- Determination of Response Factor
- A calibration curve of the various hydroxybenzene compounds is prepared by plotting the concentration of the standards versus their respective peak areas.
- Determine the Response Factor from the calibration curve.
- Sample Quantification
- The amount of the various phenolic compounds in smoke samples is quantified by the external standard method.
- The identification of peaks is by comparison of retention times with standards, and the spiking of smoke samples.
- Determination of Phenol Deliveries in μg/cigarette
- Hydroxybenzene [μg/cigarette] = [Peak Area / Resp Factor ] × [ DF / No. of Cigarettes].
where DF is the dilution factor.
- Hydroxybenzene [μg/cigarette] = [Peak Area / Resp Factor ] × [ DF / No. of Cigarettes].
15 Quality Control
- Typical Chromatogram
- See Figure 1, 2.
- Recoveries and Levels of Contamination
- Each analytical run of test cigarettes should also include:
A Laboratory Reagent Blank (LRB) to evaluate the extent of any interference due to glassware, trapping reagents, filter pads, and analyzer effects.
A Laboratory Fortified Blank (LFB) to evaluate the extent of potential analyte loss. - A standard run as a sample to verify the calculation process and validate the calibration.
- Each analytical run of test cigarettes should also include:
- Method Detection Limit (MDL) and Limit of Quantitation (LOQ)
- Method Detection Limit (MDL)
- The method detection limit is determined by analyzing the lowest level standard at least 10 times as an unknown over several days. The MDL is then calculated as three times the standard deviation of these determinations.
- Limit of Quantitation (LOQ)
- The limit of quantification is determined by analyzing the lowest level standard at least 10 times as an unknown over several days. The LOQ is then calculated as 10 times the standard deviation of these determinations.
- Method Detection Limit (MDL)
- Stability of Reagents and Supplies
- All primary stock Phenol standards are prepared fresh weekly.
- All work standards, and reagents are prepared fresh weekly.
- All samples are analyzed as soon as they are smoked and within 24 hours.
16 Modifications for Intense Smoking Conditions
- The number of cigarettes does not have to be altered for intense smoking.
References
- Risner, C.H. and Cash, S.L. "A High Performance Liquid Chromatographic Determination of Major Phenolic Compounds in Tobacco Smoke", Journal of Chromatographic Science, p. 28, 1990.
- Proctor, C.J., Martin, C., Beven, J.L., and Dymond H.F., 1988. Evaluation of an Apparatus Designed for the Collection of Sidestream Tobacco Smoke, Analyst 113: p. 1509-1513.
18 Appendices
Appendix 1: Calibration Standards
(a): Phenol Standards
This table provides a summary of the primary and secondary phenol standards for target analytes, including hydroquinone, resorcinol, catechol, phenol, o-Cresol, o-cresol, m+p-Cresol. The table contains information regarding weight of the chemical, purity, volume of the sample, as well as the stock concentrations for the primary and secondary standards, as well as for the tertiary solutions.
| Phenol | Primary (1°) Standard * | Secondary (2°) Standard * | Tertiary (3°) Solution ** | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Weight (g) | Purity (%) | Volume (mL) | Stock [mg/mL] | Vol (mL) 1 Stock | Dilute to Vol (mL) | Stock [mg/mL] | Vol (mL) Stock | Dilute to Vol (mL) | Stock [mg/mL] | |
| Hydroquinone | 0.0243 | 99.0 | 25.0 | 0.96228 | 0.50 | 50.0 | ||||
| Resorcinol | 0.0294 | 99.0 | 25.0 | 1.16424 | 2.0 | 10.0 | 0.23285 | 0.20 | 50.0 | |
| Catechol | 0.0223 | 99.0 | 25.0 | 0.88308 | 0.25 | 50.0 | ||||
| Phenol | 0.0272 | 99.0 | 25.0 | 1.07712 | 0.50 | 50.0 | ||||
| m-Cresol | 0.0335 | 99.0 | 25.0 | 1.3266 | 1.0 | 10.0 | 0.13266 | 1.00 | 50.0 | |
| p-Cresol | 0.0351 | 99.0 | 25.0 | 1.38996 | 0.5 | 10.0 | 0.06950 | 1.00 | 50.0 | |
| o-Cresol | 0.0303 | 99.0 | 25.0 | 1.19988 | 0.4 | 10.0 | 0.04800 | 2.00 | 50.0 | |
| m+p-Cresol | 99.0 | 25.0 | ||||||||
| * In 1 % (v/v) Acetic Acid ** In 1 % (v/v) Acetic Acid in a single 50 mL volumetric flask |
||||||||||
(b): Phenol Working Standards
The following table contains the volume of the working samples and the amount of chemicals in various carbonyl working standards (labelled 5, 10,100,200,350, 500, 750, 1000). These chemicals include hydroquinone, resorcinol, catechol, phenol, o-Cresol, p-cresol, m+p-Cresol.
| Label | 5 | 10 | 100 | 200 | 350 | 500 | 750 | 1000 |
|---|---|---|---|---|---|---|---|---|
| Vol (mL) 3° | 0.050 | 0.100 | 1.000 | 2.000 | 3.500 | 5.000 | 7.500 | 10.000 |
| Phenol | [μg/mL] | [μg/mL] | [μg/mL] | [μg/mL] | [μg/mL] | [μg/mL] | [μg/mL] | [μg/mL] |
| Hydroquinone | 0.04811 | 0.09623 | 0.96228 | 1.92456 | 3.36798 | 4.81140 | 7.21710 | 9.62280 |
| Resorcinol | 0.00466 | 0.00931 | 0.09314 | 0.18628 | 0.32599 | 0.46570 | 0.69854 | 0.93139 |
| Catechol | 0.02208 | 0.04415 | 0.44154 | 0.88308 | 1.54539 | 2.20770 | 3.31155 | 4.41540 |
| Phenol | 0.05386 | 0.10771 | 1.07712 | 2.15424 | 3.76992 | 5.38560 | 8.07840 | 10.77120 |
| m-Cresol | 0.01327 | 0.02653 | 0.26532 | 0.53064 | 0.92862 | 1.32660 | 1.98990 | 2.65320 |
| p-Cresol | 0.00695 | 0.01390 | 0.13900 | 0.27799 | 0.48649 | 0.69498 | 1.04247 | 1.38996 |
| o-cresol | 0.00960 | 0.01920 | 0.19198 | 0.38396 | 0.67193 | 0.95990 | 1.43986 | 1.91981 |
| m+p-Cresol | 0.02022 | 0.04043 | 0.40432 | 0.80863 | 1.41511 | 2.02158 | 3.03237 | 4.04316 |
| + In 1 % (v/v) Acetic Acid in single 10 mL volumetric flasks |
||||||||
(c): Spiking Solution
This table provides a summary of information for spiking solutions. These include the volume and concentration of both the stock and the spiked solution, as well as the analyzed concentrations for each of the following analytes: hydroquinone, phenol and o-cresol.
| Phenol | LFB Spiking Solution *** | LFM Spike ++ | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Stock Level | Stock [mg/mL] | Volume (mL) | Dilute Vol (mL) | Spike [μg/mL] | Analyzed [μg/mL] | Spike Vol (mL) | Dilute to Vol (mL) | Spike [μg/mL] | Analyzed [μg/mL] | |
| Hydroquinone | Primary | 0.96228 | 1.0 | 36.4912 | 0.76982 | 19.24560 | 1.92456 | |||
| Phenol | Primary | 1.07712 | 0.6 | 25.0 | 25.85088 | 0.51702 | 5.0 | 10.0 | 12.92544 | 1.29254 |
| o-cresol | Secondary | 0.04800 | 1.4 | 2.68773 | 0.05375 | 1.34387 | 0.13439 | |||
| *** In 1% (v/v) Acetic Acid in a single 25 mL volumetric flask ++ In 1% (v/v) Acetic Acid in a single 10 mL volumetric flask |
||||||||||
Figure 1: Chromatogram of a Typical Phenol Calibration Standard
The following displays a chromatogram for a typical phenol calibration standard.
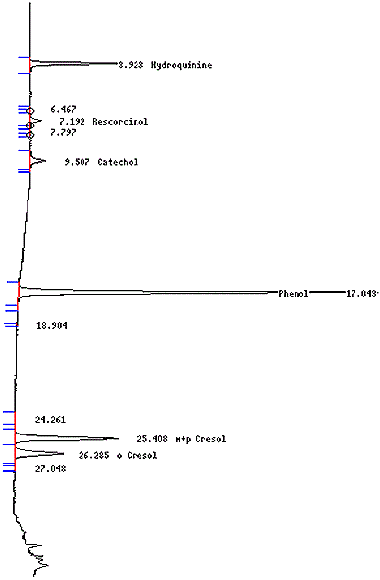
Figure 2: Chromatogram of The Analysis of Sidestream TPM for Hydroxybenzenes
The following displays a chromatogram for the sidestream total particulate matter for hydroxybenzenes.
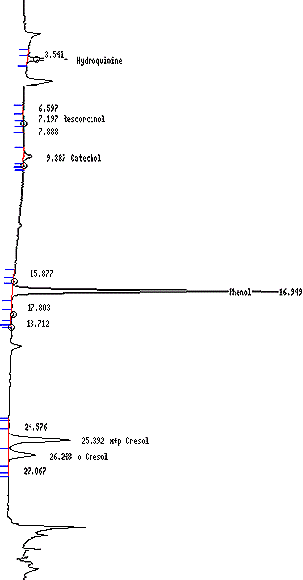
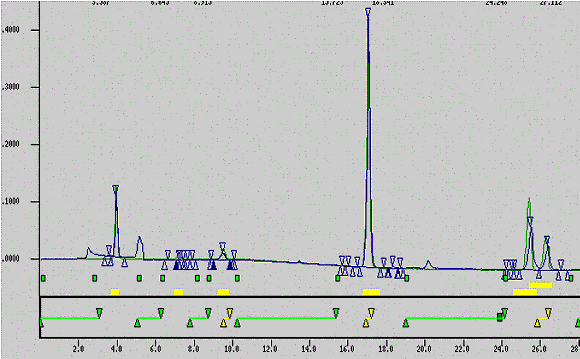
Figure 3: Overlay of Control Cigarette Phenols and Calibration Standard
The following displays an overlay of control cigarette phenols and calibration standards.
Appendix 4a: Hydroquinone Calibration Curve
The following figure displays a calibration curve for hydroquinone, where the peak size obtained by the sample was plotted against the sample concentration in ug/mL. A correlational R squared value of 0.999628 was obtained.

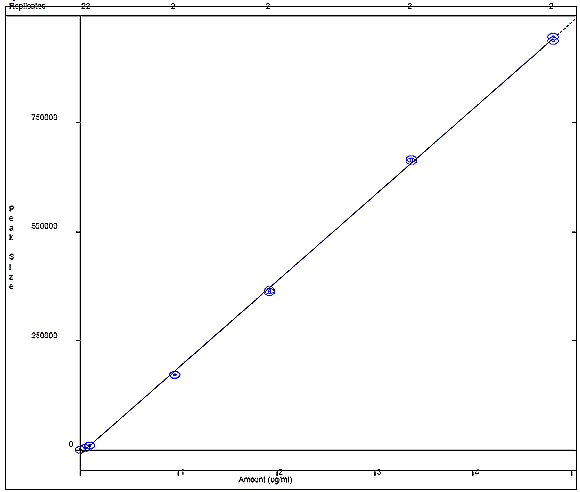
Appendix 4b: Resorcinol Calibration Curve
The following figure displays a calibration curve for resorcinol, where the peak size obtained by the sample was plotted against the sample concentration in ug/mL. A correlational R squared value of 0.998306 was obtained.

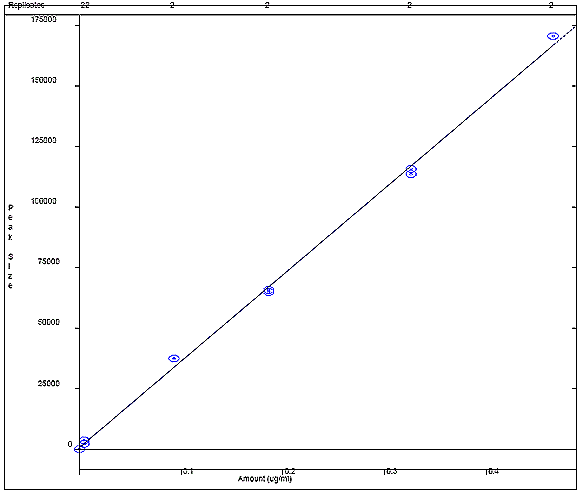
Appendix 4c: Catechol Calibration Curve
The following figure displays a calibration curve for catechol, where the peak size obtained by the sample was plotted against the sample concentration in ug/mL. A correlational R squared value of 0.998611 was obtained.

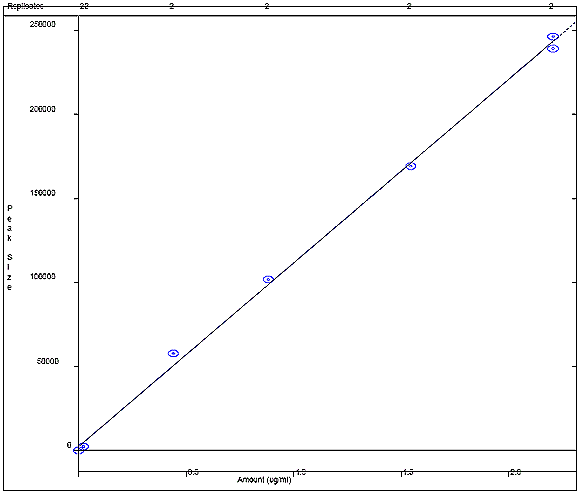
Appendix 4d: Phenol Calibration Curve
The following figure displays a calibration curve for phenol, where the peak size obtained by the sample was plotted against the sample concentration in ug/mL. A correlational R squared value of 0.999767 was obtained.


Appendix 4e: m+p Cresol Calibration Curve
The following figure displays a calibration curve for m+p cresol, where the peak size obtained by the sample was plotted against the sample concentration in ug/mL. A correlational R squared value of 0.999778 was obtained.

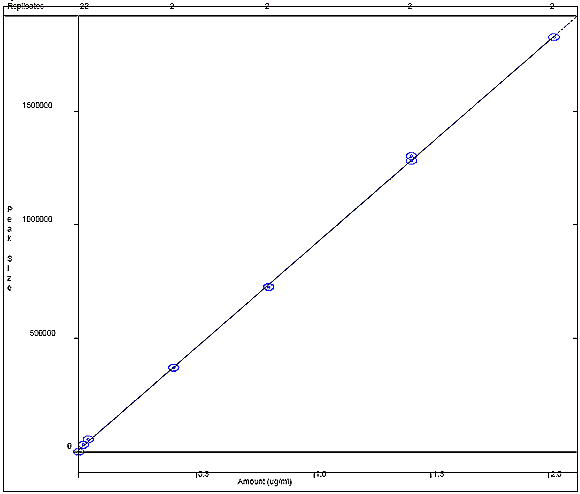
Appendix 4f: o-Cresol Calibration Curve
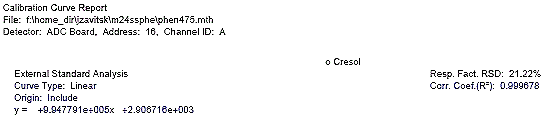
The following figure displays a calibration curve for o-cresol, where the peak size obtained by the sample was plotted against the sample concentration in ug/mL. A correlational R squared value of 0.999678 was obtained.

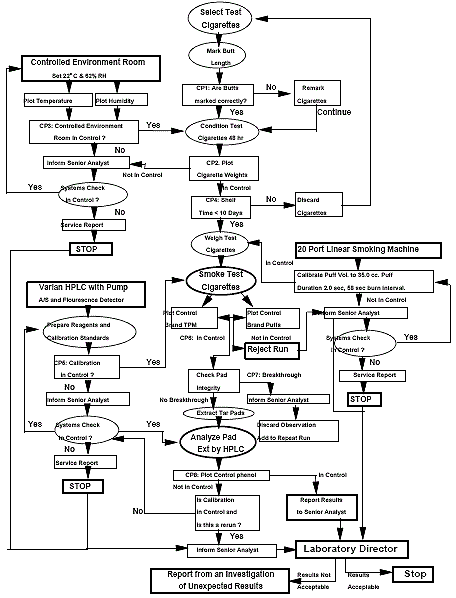
Appendix 5: Phenols Process Control Flow Diagram
The following schematic displays the phenol process control flow diagram. This maps out the systematic work flow for processing phenol in the system, ranging from laboratory tests to generating service reports.
Appendix 6a: Laboratory Reagent Blanks (LRB) from a Recent Sidestream Study
The following table displays the laboratory fortified blanks (LFB) from a recent sidestream study; results include the initial concentration of the following chemicals: hydroquinone, resorcinol, catechol, phenol, m+p cresol, o-cresol. Three samples were analyzed over 3 days. The table also displays the corresponding average percent laboratory reagent blank concentrations for each chemical.
| Sample Description | Hydroquinoine (ug/ml) | Resorcinol (ug/ml) | Catechol (ug/ml) | Phenol (ug/ml) | m+p Cresol (ug/ml) | o-Cresol (ug/ml) |
|---|---|---|---|---|---|---|
| Day1_LRBa | Not Det | 0.0112 | 0.0260 | 0.0113 | 0.0218 | 0.0003 |
| Day2_LRBa | Not Det | 0.0107 | 0.0235 | 0.0113 | Not Det | 0.0076 |
| Day3_LRBa | Not Det | 0.0139 | 0.0468 | 0.0136 | Not Det | 0.0044 |
| Sample Description | Hydroquinoine (ug/cig) | Resorcinol (ug/cig) | Catechol (ug/cig) | Phenol (ug/cig) | m+p Cresol (ug/cig) | o-Cresol (ug/cig) |
|---|---|---|---|---|---|---|
| Day1_LRBa | Not Det | 0.112 | 0.260 | 0.113 | 0.218 | 0.003 |
| Day2_LRBa | Not Det | 0.107 | 0.235 | 0.113 | Not Det | 0.076 |
| Day3_LRBa | Not Det | 0.139 | 0.468 | 0.136 | Not Det | 0.044 |
| Average | Not Det | 0.119 | 0.321 | 0.121 | 0.218 | 0.041 |
Appendix 6b: Laboratory Fortified Blanks (LFB) from a Recent Sidestream Study
Results are reported on a Per cent Recovered Basis
The following table displays the laboratory fortified blanks (LFB) from a recent side stream study, results include the initial concentration of hydroquinone, phenol , and o-cresol for three samples collected over 3 days, as well as the corresponding average percent laboratory reagent blank. Recoveries for each chemical are recorded in a percent recovery basis.
| Sample Description | Hydroquinoine (ug/ml) | Phenol (ug/ml) | o-Cresol (ug/ml) |
|---|---|---|---|
| Day1_LRBa | 73.40 | 94.00 | 114.23 |
| Day2_LRBa | 83.10 | 96.75 | 74.23 |
| Day3_LRBa | 90.23 | 97.25 | 125.21 |
| Average | 82.24 | 96.00 | 104.56 |
Appendix 6c: Laboratory Fortified Matrix (LFM) from a Recent Sidestream Study
Results are reported on a Per Cent Recovered Basis
The following table displays the laboratory fortified matrix (LFM ) from a recent side stream study, results include the initial concentration of hydroquinone, phenol, and o-cresol for three samples, as well as the corresponding average percent laboratory fortified matrix. Recoveries for each chemical are recorded in a percent recovery basis.
| Sample Description | Hydroquinoine (ug/ml) | Phenol (ug/ml) | o-Cresol (ug/ml) |
|---|---|---|---|
| R04_P12_LFMa | 98.76 | 93.75 | 95.84 |
| R08_P09_LFMa | 99.02 | 102.82 | 102.76 |
| R12_P17_LFMa | 100.78 | 109.32 | 124.93 |
| Average | 99.52 | 101.96 | 107.85 |
Appendix 7: Minimum Detection Limit (MDL) and Limit of Quantitation (LOQ) for
Sidestream Phenols
The following table displays the mm detection limit (MDL) and limit of quantitation (LOQ) for the concentration of sidestream phenols found in various chemical standards, including hydroquinone, resorcinol, catechol, phenol, m+p cresol and o-cresol. The average, standard deviation and coefficient of variance were found among all samples.
| Phenols | Standard 1 Hydroquinoine (ug/ml) |
Standard 2 Resorcinol (ug/ml) |
Standard 2 Catechol (ug/ml) |
Standard 1 Phenol (ug/ml) |
Standard 2 m+p Cresol (ug/ml) |
Standard 2 o-Cresol (ug/ml) |
|---|---|---|---|---|---|---|
| 0.0512 | 0.0092 | 0.0543 | 0.0427 | 0.0355 | 0.0215 | |
| 0.0510 | 0.0069 | 0.0598 | 0.0474 | 0.0342 | 0.0193 | |
| 0.0493 | 0.0052 | 0.0557 | 0.0465 | 0.0356 | 0.0229 | |
| 0.0514 | 0.0071 | 0.0619 | 0.0474 | 0.0354 | 0.0211 | |
| 0.0490 | 0.0068 | 0.0570 | 0.0484 | 0.0351 | 0.0179 | |
| 0.0543 | 0.0054 | 0.0506 | 0.0489 | 0.0368 | 0.0182 | |
| 0.0544 | 0.0056 | 0.0439 | 0.0424 | 0.0366 | 0.0202 | |
| 0.0497 | 0.0059 | 0.0472 | 0.0424 | 0.0357 | 0.0205 | |
| 0.0506 | 0.0071 | 0.0485 | 0.0451 | 0.0353 | 0.0221 | |
| 0.0538 | 0.0073 | 0.0628 | 0.0452 | 0.0347 | 0.0189 | |
| Average | 0.0515 | 0.0067 | 0.0542 | 0.0456 | 0.0355 | 0.0203 |
| Std Dev | 0.0020 | 0.0012 | 0.0065 | 0.0025 | 0.0008 | 0.0017 |
| Coeff of Var | 3.9 | 17.9 | 11.9 | 5.4 | 2.2 | 8.3 |
| MDL (ug/ml) | 0.0061 | 0.0036 | 0.0194 | 0.0074 | 0.0023 | 0.0050 |
| MDL (ug/cig) | 0.122 | 0.071 | 0.387 | 0.149 | 0.047 | 0.100 |
| LOQ (ug/ml) | 0.0203 | 0.0119 | 0.0646 | 0.0248 | 0.0078 | 0.0167 |
| LOQ (ug/cig) | 0.406 | 0.238 | 1.291 | 0.496 | 0.156 | 0.335 |
Appendix 8: Kentucky Reference cigarette (1R4F) Yields
Sidestream Yield Summary for Kentucky Reference 1R4F (Brand 507)
The following table displays the sidestream yield summary of kentucky reference cigarette 1R4F (Brand 507). This table provides the mean concentration and standard deviation found for the following analytes: hydroquinone, resorcinol, catechol, phenol, mp_cresol and o_cresol.
| Analyte Phenolic grp | Mean | Units | Std. Dev. | Coeff. Var. |
|---|---|---|---|---|
| Hydroquinone | 116 | ug/cig | 17.3 | 14.9% |
| Resorcinol | 0.806 | ug/cig | 0.568 | 70.5% |
| Catechol | 93.4 | ug/cig | 13.9 | 14.9% |
| Phenol | 247 | ug/cig | 25.7 | 10.4% |
| mp_cresol | 72.9 | ug/cig | 10.1 | 13.8% |
| o_cresol | 34.7 | ug/cig | 5.82 | 16.8% |